Background We conducted a population-based, retrospective study to examine the risk of a variety of connective-tissue diseases and other disorders after breast implantation.
Methods All women in Olmsted County, Minnesota, who received a breast implant between January 1, 1964, and December 31, 1991 (the case subjects), were studied. For each case subject, two women of the same age (within three years) from the same population who had not received a breast implant and who underwent a medical evaluation within two years of the date of the implantation in the case subject were selected as control subjects. Each woman's inpatient and outpatient medical record was reviewed for the occurrence of various connective-tissue diseases, certain other disorders thought to have an autoimmune pathogenesis (e.g., Hashimoto's thyroiditis), and cancer other than breast cancer, as well as related symptoms and abnormal results of laboratory tests. The case subjects were categorized according to whether they received implants for cosmetic reasons, for reconstruction after mastectomy for breast cancer, or for reconstruction after subcutaneous mastectomy for cancer prophylaxis. Additional control subjects (women treated for breast cancer who did not have breast reconstruction) were studied for comparison with the case subjects.
Results A total of 749 women who had received a breast implant were followed for a mean of 7.8 years, and 1498 community controls were followed for a mean of 8.3 years. In 5 case subjects, as compared with 10 subjects in the control group, one of the specified connective-tissue diseases was diagnosed (relative risk, 1.06; 95 percent confidence interval, 0.34 to 2.97). Twenty-five case subjects had signs or symptoms of arthritis, as compared with 39 control subjects (relative risk, 1.35; 95 percent confidence interval, 0.81 to 2.23). Among the various signs or symptoms examined, only morning stiffness was significantly increased among the women who had received a breast implant (relative risk, 1.81; 95 percent confidence interval, 1.11 to 2.95).
Conclusions We found no association between breast implants and the connective-tissue diseases and other disorders that were studied. .
We defined connective-tissue disease as a group of disorders that includes rheumatoid arthritis, systemic lupus erythematosus, Sjogren's syndrome, dermatomyositis, polymyositis, systemic sclerosis, ankylosing spondylitis, psoriatic arthritis, polymyalgia rheumatica, vasculitis, arthritis associated with inflammatory bowel disease, and polychondritis. Other disorders considered included Hashimoto's thyroiditis, primary biliary cirrhosis, sarcoidosis, and cancer other than breast cancer.
Methods
Study Setting
Olmsted County is relatively isolated from other urban areas. Medical care is concentrated among a handful of providers, one of which, the Mayo Clinic, is also a major tertiary care center. A unit record system allows all medical information on each person to be accumulated in a single dossier14. Mayo has maintained the original records of every patient seen since 1907 (more than 4.3 million patients to date). The records can be accessed by indexes to the clinical and pathological diagnoses and surgical procedures that are incorporated in what is known as the Rochester Epidemiology Project. Thus, medical records from essentially all sources of health care available to residents of the county are linked.
In any given year, over half the residents of the county receive medical care at the Mayo Clinic (or its affiliates, Saint Mary's Hospital and Rochester Methodist Hospital), and another 20 to 25 percent are treated at the Olmsted Medical Group (or its affiliate, Olmsted Community Hospital). The records in the Rochester Epidemiology Project include all medical care provided at these facilities under the supervision of a physician, except some private psychiatry services. In a recent survey of 2122 Rochester residents identified by random-digit dialing, every resident had a medical record at the Mayo Clinic, the Olmsted Medical Group, or the Olmsted Community Hospital15. We have estimated that 95 percent or more of the local residents (depending on age) will visit at least one of these facilities in a three-year period.
Identification of Case and Control Subjects
All county residents whose medical records indicated that they had received a breast implant between January 1, 1964, and December 31, 1991, were included as case subjects. For each of these women, two women of the same age (within three years) who had undergone a medical evaluation within two years of the date of breast implantation in the case subject were selected from among county residents. Among the eligible control subjects, the ones chosen were those with the registration numbers nearest to those of the case subjects. Since unique registration numbers are assigned at the time of the initial registration for care, this method matches a case subject with two control subjects according to the duration of prior medical care in the county. In prior studies, most control subjects selected in this way were persons who had received routine or minor medical care. The case subjects were classified according to the indication for breast implantation -- i.e., cosmetic reasons, reconstruction after mastectomy for breast cancer, or reconstruction after subcutaneous mastectomy for cancer prophylaxis among women at high risk for breast cancer.
For each woman receiving an implant after a mastectomy for breast cancer, two additional county residents who had undergone mastectomy for breast cancer but did not have a breast implant were selected as control subjects. Thus, there were four control subjects for each case subject who had been treated for breast cancer. The primary analysis was performed with control subjects from the community. This study was limited to women living in the county on the index date (the date of implantation for the case subjects, the date of the closest medical visit for the control subjects, and the date of surgery for women who underwent mastectomy for breast cancer but did not receive an implant). Follow-up of all women continued from the index date until death, December 31, 1991, or the date of the woman's last health care visit in the county.
Outcomes
For each woman studied, the entire medical record, including all inpatient and outpatient reports by any local provider of medical care, was reviewed by one of four trained nurses who sought evidence of the clinical features, laboratory studies, and radiographic studies necessary to make the following diagnoses: systemic sclerosis, systemic lupus erythematosus, rheumatoid arthritis, polymyositis, dermatomyositis, Sjogren's syndrome, keratoconjunctivitis sicca, lymphoproliferative disorders, Hashimoto's thyroiditis, primary biliary cirrhosis, and cancer other than breast cancer. The specific diagnostic criteria are described elsewhere16,17,18,19. Pertinent clinical and laboratory data were recorded on a pretested data-collection form, entered into a computer, and edited with a variety of on-line checks for consistency and appropriateness of range. The principal investigator and the study coordinator met weekly with the nurses reviewing the records to resolve questions about the interpretation of entries and to monitor adherence to the protocol.
Statistical Analysis
The case and control groups were matched according to age and year of implant or visit. Since no differences in results were observed between matched and unmatched analyses, only the results of unmatched analyses are reported. The primary analysis compared the incidence of various end points in the case and control subjects over the person-years of observation, using a modified F-test developed by Cox20 that assumes that the observed events follow a Poisson distribution. The Cox proportional-hazards model21 of time to the occurrence of an end point and the generalized linear model (with log-link function)22 of the incidence of events over person-years of observation were used to compare the rates of end points in the case subjects with those in the control subjects, with adjustment for covariates. Both methods allow for the inclusion of covariates in the models to adjust for their influence on rates and on the comparison of case with control subjects. Age and index year were included in all analyses. All statistical tests were two-tailed, and a P value of <0.05 was considered to indicate statistical significance.
Results
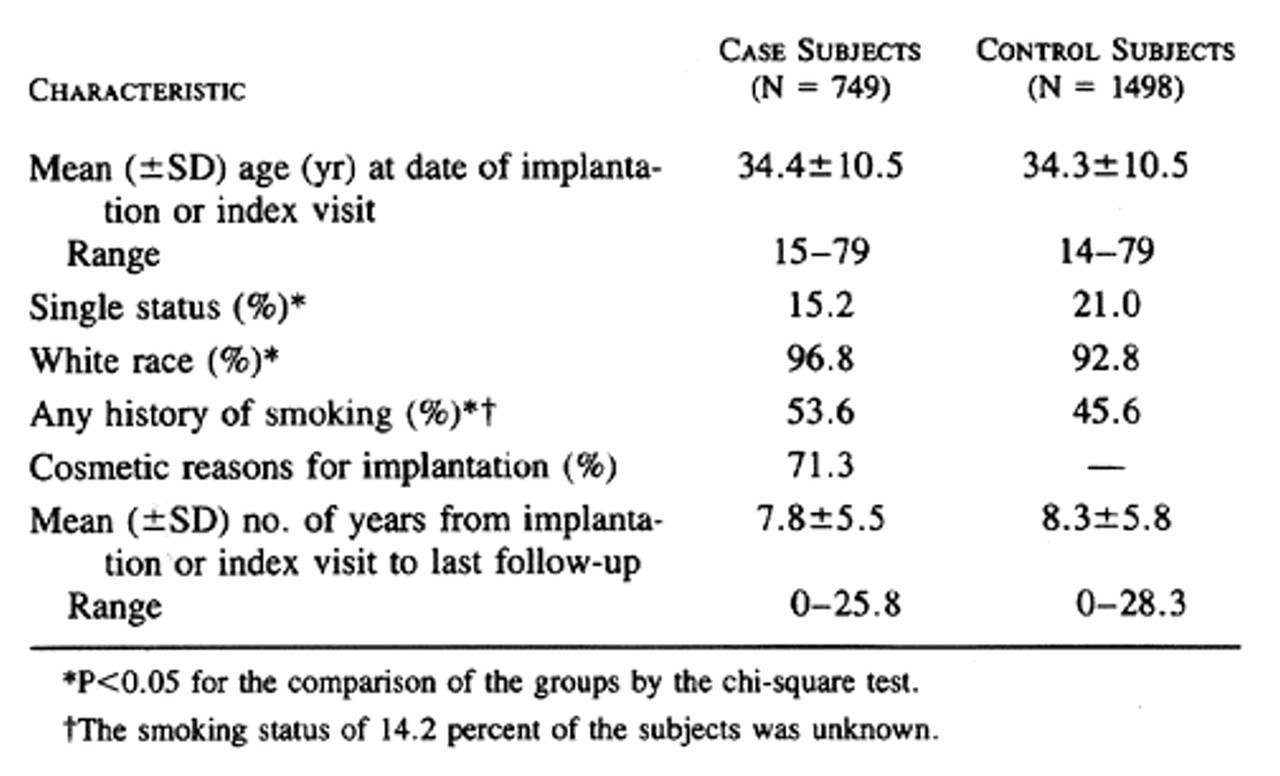
The 749 county residents who received a first breast implant between January 1, 1964, and December 31, 1991 (the case subjects), were followed for 5847 person-years (mean, 7.8 years per case subject). The control subjects from the community included 1498 women who were followed for 12,361 person-years (mean, 8.3 years per control subject). The control subjects with cancer included 306 women with breast cancer who did not receive a breast implant and who were followed for 2703 person-years (mean, 8.8 years per subject). There was little difference in mean age or duration of follow-up between the case and control subjects , but marital status, ethnic background, and smoking history differed significantly.
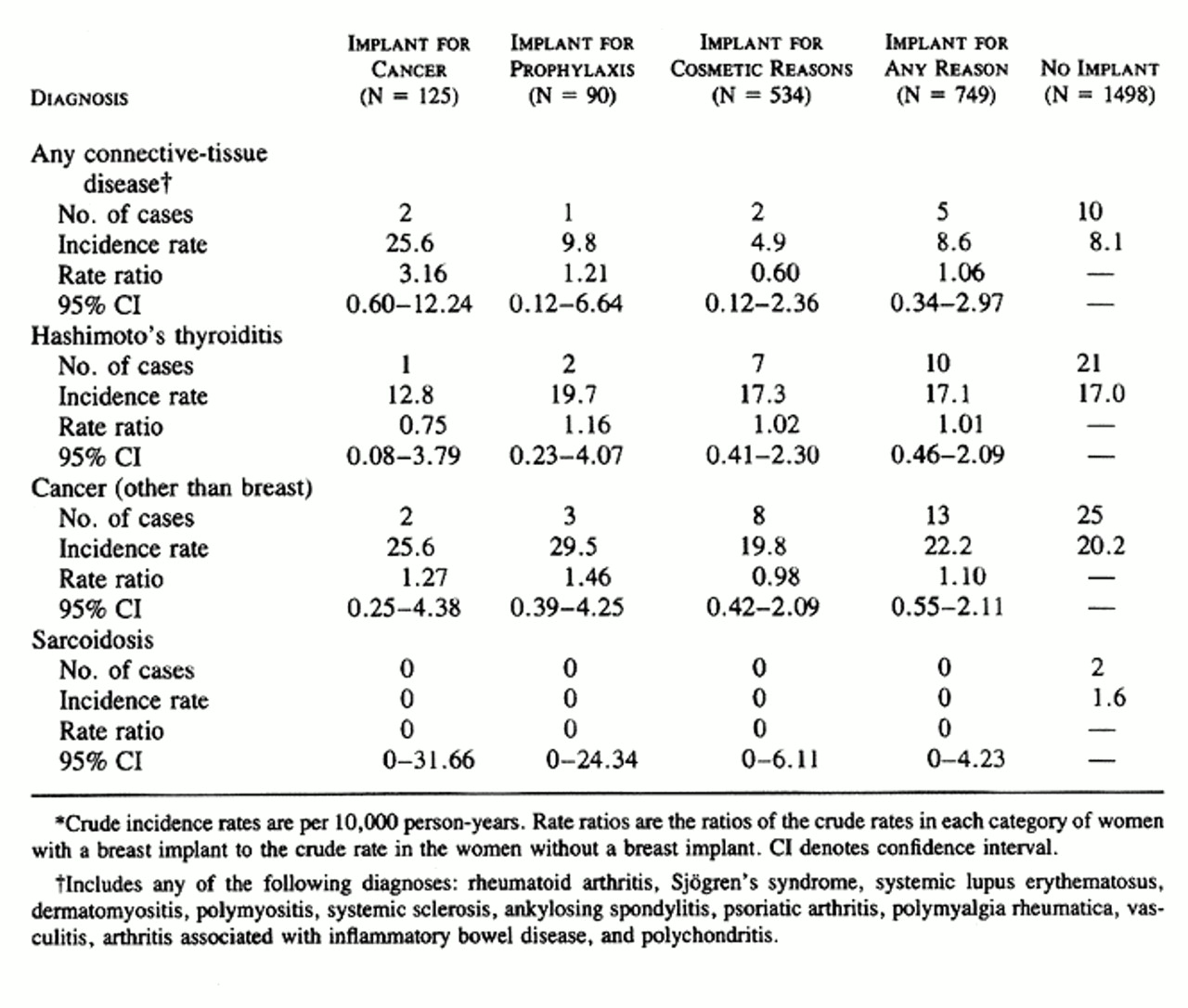
The case subjects were compared with the control subjects with regard to the occurrence of clinical signs and symptoms of the connective-tissue diseases and other disorders being studied, abnormal laboratory tests suggestive of these conditions, and diagnoses of these diseases. Five of the 749 case subjects had been given a diagnosis of connective-tissue disease (polymyalgia rheumatica in 2 and Sjogren's syndrome, polychondritis, and arthritis associated with inflammatory bowel disease in 1 each), as compared with 10 of the 1498 community controls (3 with ankylosing spondylitis, 2 each with rheumatoid arthritis and systemic vasculitis, and 1 each with psoriatic arthritis, polymyalgia rheumatica, and systemic sclerosis with polyarthritis) (relative risk, 1.06; 95 percent confidence interval, 0.34 to 2.97).
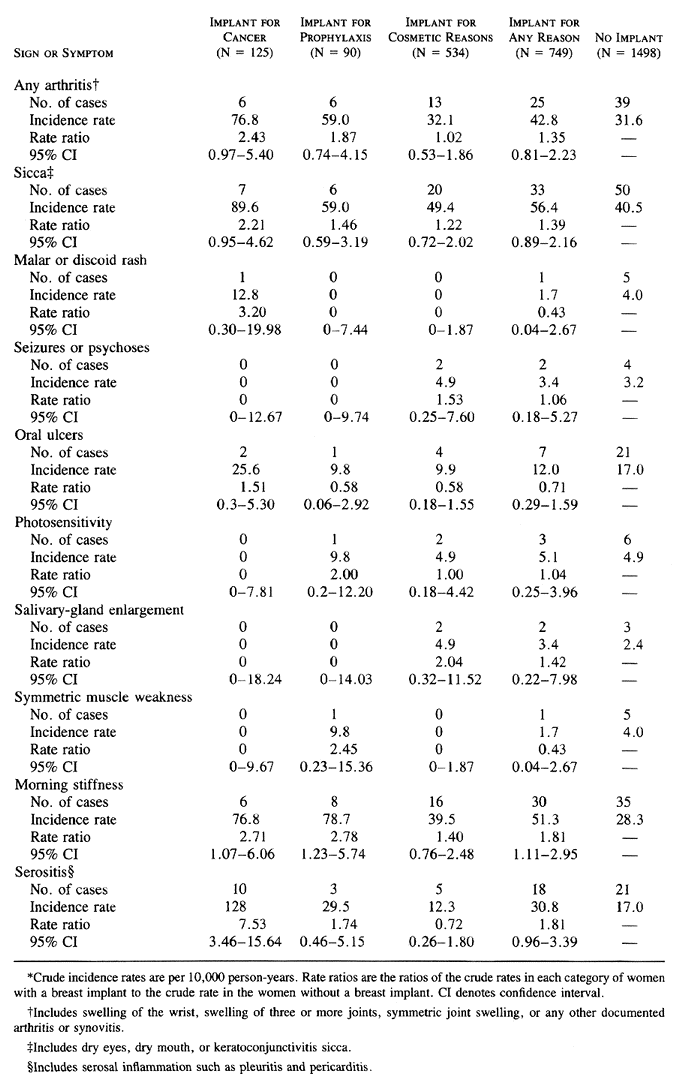
No significant differences were found between the case and control subjects in the relative risk of abnormal results on laboratory testing for antinuclear antibody, rheumatoid factor, or antimicrosomal antibody (Table 4). Abnormal results of the laboratory test for thyroid-stimulating hormone were significantly more frequent among the control subjects than among the case subjects (Table 4).

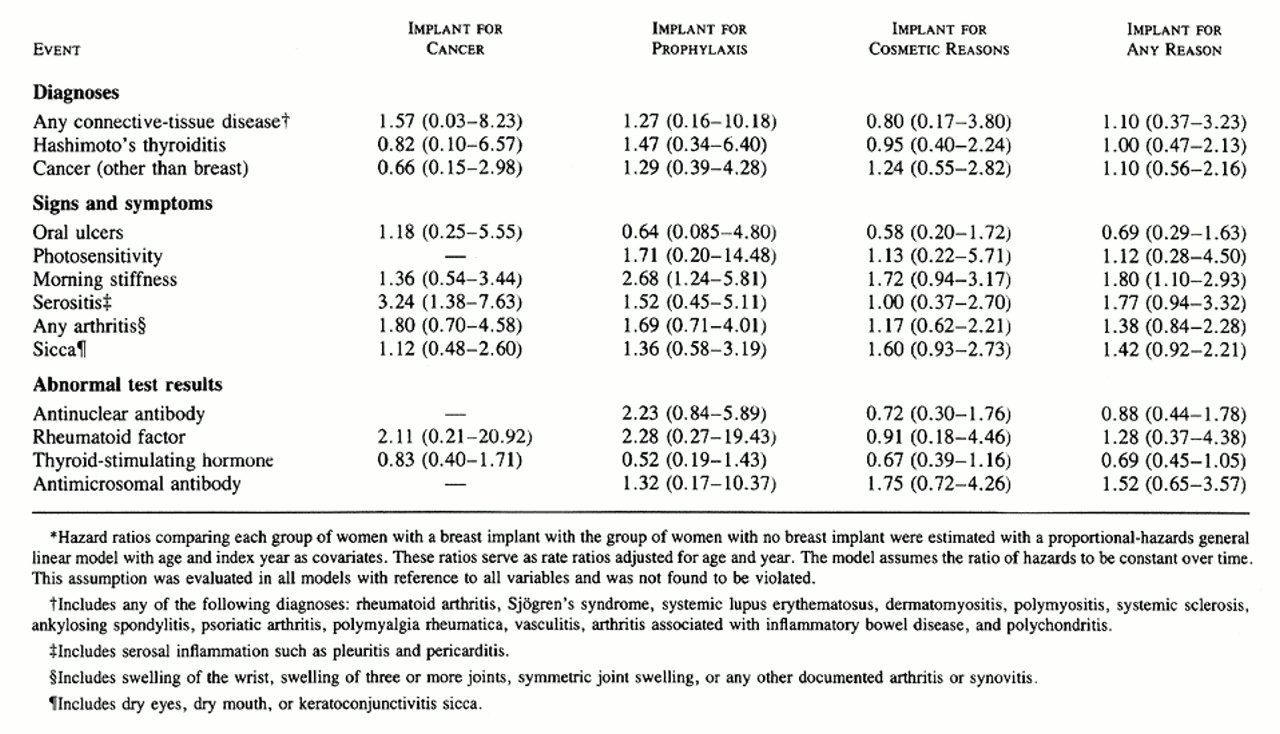
The comparisons in Table 2, Table 3, and Table 4 did not involve adjustment for age or index year, the two variables used in matching the case subjects and the control subjects, or for ethnic background, marital status, or smoking history, variables on which the two groups differed ( Table 1). In the case of end points that occurred often enough to warrant a more complex analysis, the Cox proportional-hazards model was used to obtain estimates of hazard ratios adjusted for age and index year ( Table 5). The other variables were not included, because they either had no effect on the results and were never significant (marital status and smoking history) or were rare (race other than white). Although the majority of these hazard ratios were very similar to the rate ratios, there were some differences, presumably due to the adjustment for age. As in the unadjusted analysis, the only significantly elevated risks in the proportional-hazards analysis were those for serositis, in the women who underwent breast implantation after mastectomy for breast cancer, and for morning stiffness, among all women with implants and those who had implants after subcutaneous mastectomy for cancer prophylaxis.
Reports of connective-tissue diseases and other disorders3,4,5,6,7,8,9,10,11,12,23,24,25,26,27,28,29,30 among women with breast implants have raised the question of a cause-and-effect relation. We believe that a definitive answer to this question would require a well-conducted prospective cohort study using a multicenter controlled design with follow-up of a large population of women for at least 10 years. Such a study would be very expensive and time-consuming. Instead, we conducted a retrospective cohort study using the extensive data resources of the Rochester Epidemiology Project.
Other studies have addressed the question of whether silicone-containing breast implants cause connective-tissue disorders31,32,33,34,35. However, these studies were limited by substantial methodologic flaws, including uncontrolled, retrospective designs,31 lack of objective validation of the survey replies,31 very small samples, inadequate statistical power, large losses to follow-up (nearly 75 percent of the initial sample in one study),31 and the use of referral-based cohorts31,32,33,34,35. In contrast, our controlled study allowed extensive follow-up of a population-based cohort of women with silicone-containing breast implants and the corresponding control group.
We found no statistically significant elevation in the relative risk of any of the specified connective-tissue diseases or other disorders among the women with breast implants as compared with the control subjects. Two conditions, morning stiffness and serositis, were significantly more frequent among the subgroup of the case subjects who received their implant after mastectomy for breast cancer (Table 5), but our data suggest that these events are more likely to be related to the underlying cancer than to the implant itself, because the incidence rates of these events were similar to those among the control women with breast cancer who did not receive a breast implant after mastectomy.
Although our results do not support the hypothesis that women with breast implants have an increased risk of connective-tissue diseases or other disorders, our study has several limitations. First, we had limited power to detect an increased risk of rare connective-tissue diseases, such as systemic sclerosis. Indeed, we calculated that it would require a sample of 62,000 women with implants and 124,000 women without implants, followed for an average of 10 years each, for a doubling of the relative risk of this condition to be detected among women with implants, assuming that the annual incidence of systemic sclerosis is 1.6 cases per 100,000 women36. Our results, therefore, cannot be considered definitive proof of the absence of an association between breast implants and connective-tissue disease. Other controlled studies of this question are ongoing. Their results, together with ours, will help to resolve this controversy.
The use of data bases linked to medical records, such as the Rochester Epidemiology Project, presents a number of potential problems37. For a disorder to be recorded in the data base, the condition must be recognized by a clinician, recorded accurately, and retrieved. Because we screened and reviewed the symptoms and laboratory results directly, events could be included when they met our definitions, even if the attending physician failed to make (or record) the diagnosis. For example, we found that contrary to previous reports,30 the incidence of abnormal test results for antinuclear antibodies in women with breast implants did not differ significantly from the incidence in women without implants in whom the test was performed. Still, conditions that are unlikely to come to medical attention may not be detected in such a data set -- for example, some episodes of "dry eyes" that could have been related to sicca syndrome may have been missed. However, we reviewed each subject's complete medical record covering an average of eight years, and it is unlikely that serious or persistent problems, such as Sjogren's syndrome or cancer, went unrecorded by every attending physician.
Our study had a long average follow-up, lasting at least 10 years for 36 percent of the cohort. Nevertheless, very late outcomes, occurring more than 10 years after surgery, could not be evaluated adequately. It may be desirable to extend the follow-up of this cohort. Finally, some racial and ethnic groups are underrepresented in Olmsted County, where the population was 96 percent white in 1990. Conditions that occur preferentially in other groups could not be assessed. However, most previous reports linking breast implants and connective-tissue disease relate to white women, and 97 percent of our case subjects were white. Thus, the results of our population-based study are more likely to apply to whites in general than are the findings from investigations of referral-based cohorts. In conclusion, our results do not support an association between breast implants and connective-tissue diseases or other disorders that were studied.
Supported by research grants from the Plastic Surgery Educational Foundation and by a grant (AR30582) from the National Institutes of Health.
We are indebted to Susan Greteman, B.S., and Joanne Benson, B.A., for performing the statistical analysis, to Beth Ryan and Lori Norby for assistance in the preparation of the manuscript, and to Joseph Duffy, M.D., for providing the idea from which this study developed.
Source Information
From the Division of Rheumatology and Internal Medicine (S.E.G.), the Department of Health Sciences Research (S.E.G., W.M.O., L.T.K., C.M.B., L.J.M.), and the Department of Surgery (J.E.W.), Mayo Clinic and Mayo Foundation, Rochester, Minn.
References